Regenerative medicine is profoundly transforming our relationship with healthcare. As a result, the human body is no longer a static therapeutic target; it is becoming a field for iterative engineering. For medtech actors and leading researchers, we are witnessing the collapse of the boundary between the living and the machine. This is no longer a simple evolution, but the birth of a circular value chain where predictive data orchestrates organic matter. This convergence between biology and artificial intelligence is part of a broader transformation of medtech, already visible in advances in AI medical diagnosis.
The origins: the architecture of cellular code
It all begins with the liberation of the cell. Under Cellink’s leadership, 3D bioprinting and biofabrication are emerging from niche laboratories to become a standardized production unit. With consolidated revenue approaching 1.5 billion SEK in 2025, the BICO Group does not sell printers, but rather a living design infrastructure. By mastering the rheology of bio-inks, medical 3D bioprinting enables experts to design tissue microarchitectures whose biomimetic complexity is the first step toward rejection-free surgery. This sub-millimeter precision builds on the advancements driven by modern regenerative medicine and medical 3D bioprinting technologies, which now form the foundation of tissue engineering.
Mapping: The Intelligence Behind Silent Navigation
Uncontrolled movement poses a risk. Brainlab has established itself as the digital conductor of this odyssey. By transforming the operating room into a spatialized environment—with more than 6,000 systems deployed worldwide by 2025—the German firm ensures continuity between diagnosis and procedure. For decision-makers, the ROI here is invisible but massive: a drastic reduction in reoperations thanks to sub-millimeter precision guided by the digital twin. This technological orchestration paves the way for the integration of regenerative medicine therapies into next-generation operating rooms.

Brainlab navigation systems allow surgeons to visualize brain structures in real time to improve surgical precision. © Brain Lab
The breakthrough: the transition to surgical autonomy
Caranx Medical has now eliminated the bottleneck posed by human hands. Affluent Medical’s strategic acquisition of the company in 2025 for €16.6 million marks a turning point: the introduction of generative AI into real-time robotic guidance. By obtaining FDA approval for its AiiR autonomous navigation software, Caranx Medical proves that the future of surgery lies not in assistance, but in the secure delegation of complex procedures. This secure delegation of complex procedures represents a key step in the industrialization of regenerative medicine.
Fusion: Trauma-free molecular adhesion
The history of surgery has been one of necessary aggression in the name of repair. Tissium puts an end to this paradox. Their light-activated synthetic polymers—backed by €50 million in funding—replace mechanical sutures with seamless biological integration. This sealing step is the cornerstone of the regenerative medicine value chain: it allows Cellink tissues to be secured within the framework defined by Brainlab, without causing a peripheral inflammatory reaction.
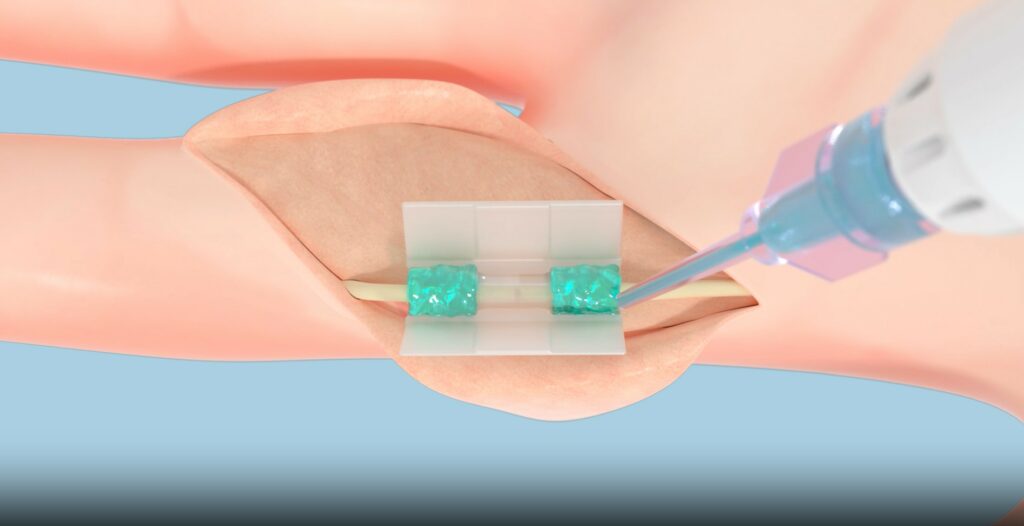
Tissium polymers enable tissue repair without traditional mechanical sutures. The viscous pre-polymer is activated on-demand, using a visible blue light. The resulting elastomeric bond ensures fixation, allowing the polymer to comply with the underlying tissue while remaining strongly fixed © Tissium
The neuro-symphony of movement
Organic repair is only valuable if it is functional. Cyberdyne provides the kinematic solution. With a 20% increase in its HAL device rental services projected for 2025, the Japanese giant is transforming brain intent into physical movement. By capturing bioelectrical signals, the exoskeleton becomes a natural extension of the repaired body, re-teaching the nervous system to inhabit its new biological architecture. Functional restoration thus becomes the natural extension of innovations in regenerative medicine. It complements the technological ecosystem where 3D bioprinting, robotics, and AI converge to restore biological functions.
Cyberdyne HAL exoskeleton detects bioelectric signals from the brain to assist movement and support neurological rehabilitation. © Cyberdyne
The sentinel of phenotypic algorithms
The cycle concludes with infrared monitoring. Median Technologies, with an order backlog of €76.6 million, deploys its AI algorithms to monitor the invisible. By analyzing the very texture of regenerated tissues, their eyonis® platform detects subtle signs of failure long before a clinical alert is triggered. This represents the final quality assurance step in a value chain where every stage of regenerative medicine is validated by data. This algorithmic validation is becoming essential for ensuring the safety of therapies derived from 3D bioprinting.
Epilogue: the silence of the scalpel
We are moving from the era of heroic medicine to that of discreet engineering. In this new world, surgery is no longer visible; it is programmed. Technology fades into the background in favor of a restored life, where the only trace of the machine’s intervention is, paradoxically, a perfect return to nature.
However, this mastery of organic matter raises a final frontier: that of identity. If every component of our architecture becomes replaceable and updated by data streams, at what point do we cease to be a biological entity and become a product of continuous engineering? The next step in this value chain may no longer be to repair the human body, but to redefine its finality.
Regenerative medicine is no longer a theoretical prospect; it is now establishing itself as an industrial infrastructure for living organisms, profoundly redefining the foundations of contemporary medtech. At the heart of this transformation, 3D bioprinting is emerging as one of the major technological pillars of regenerative medicine.




















